What is brown fat (brown adipose tissue) ?
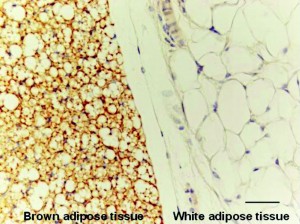 Beta-lapachone and brown fat cell targeting offers great hope for those struggling with age related diseases. The adipose organ in mammals is composed of both white and brown adipose tissue. Brown adipose tissue, or BAT, is especially present in newborn infants and hibernating mammals. Brown fat deposits, often referred to as baby fat, tend to decline in humans as they age. The primary metabolic function of brown fat is thermoregulation. Brown fat stimulates energy expenditure by producing heat through both shivering and non-shivering thermogenesis. The heat is produced via the electron transport chain.
Beta-lapachone and brown fat cell targeting offers great hope for those struggling with age related diseases. The adipose organ in mammals is composed of both white and brown adipose tissue. Brown adipose tissue, or BAT, is especially present in newborn infants and hibernating mammals. Brown fat deposits, often referred to as baby fat, tend to decline in humans as they age. The primary metabolic function of brown fat is thermoregulation. Brown fat stimulates energy expenditure by producing heat through both shivering and non-shivering thermogenesis. The heat is produced via the electron transport chain.
Brown adipocytes contain iron rich mitochondria and numerous small lipid droplets. In contrast, white adipocytes contain only one lipid droplet and fewer iron containing mitochondria. Brown fat is more efficient than white fat at distributing heat, delivering nutrients and providing oxygen to the body. This is primarily due to the fact that brown fat is equipped with more capillaries than white fat.
The primary brown fat deposits in humans occur around the neck, kidneys, spinal cord and between the shoulder blades. The human body also features beige fat, which displays variable lipid droplet profiles and is dispersed throughout the body. Beige adipose tissue is also characterized by a greater lipid droplet to mitochondria ratio than brown fat.
Hair Restoration
Hair loss researchers and hair growth products have focused almost exclusively on blocking dihydrotestosterone, or DHT, in recent decades. Although there are many factors that contribute to androgenetic alopecia, or male pattern baldness, recent research suggests that factors such as decreased subcutaneous fat, inadequate follicle microcirculation, miniaturized follicles and localized inflammation may actually be to blame.1
It appears that brown adipose tissue plays an important role in protecting hair follicles from miniaturization and hair loss. Brown fat is a subcutaneous layer of fat deep within the skin. Brown fat, or baby fat, decreases with age, especially in men. Men that suffer from androgenetic alopecia, particularly older and overweight men, have reduced levels of brown fat.
Research indicates that productive hair follicles are embedded in brown fat. Dormant hair follicles and balding areas typically exhibit little or no brown adipose tissue. The back of the head, especially the lower part of the occipital region, is far more likely to have brown adipose tissue in the subcutaneous depths of the skin. That’s why many androgenetic alopecia sufferers still enjoy thick hair growth on the back of the head. Follicle miniaturization and hair loss does not occur until the brown adipose tissue begins to decline. Simply put, brown fat is a vital substrate that promotes follicle health and normal hair syntheses.
Loss of brown adipose tissue is caused by inadequate microcirculatory blood flow. Blood vessel restriction can be caused by a variety of factors, including skull expansion, nutritional deficiencies, harmful chemicals, heat from hair dryers, excessive hair cutting and autoimmune inflammation. Brown fat deposits support normal hair growth by promoting blood vessel formation, providing a cushion to minimize the effects of skull expansion and the availability of an environment in which hair follicles can penetrate deeply into the dermis to obtain an adequate blood supply.
Increasing brown fat levels, therefore, will revitalize dysfunctional follicles and promote normal hair growth. Healthy hair follicles thrive in a scalp that is flexible and delivers maximum blood vascularization.
Aging and Obesity
Brown fat is dedicated to the survival of homeotherms and maintaining body temperature. Brown adipose tissue is also essential for thermoregulation due to its unique role in the dissipation of stored energy.3 Brown fat researchers are searching for therapies to treat obesity, cancer, metabolic syndrome and other diseases by restoring brown fat levels in the body. The discovery of mechanisms that restore normal brown fat mediated thermogenesis and temperature homeostasis have the potential to effectively treat many chronic diseases.
Although it was assumed that brown adipose tissue disappears after birth, aging researchers have employed new functional imaging to confirm that brown fat activity is present at all stages of life. Restoration of normal brown fat levels may prove useful in the treatment of many age related diseases. Researchers believe that PET tomography underestimates the prevalence of brown fat in human adults since PET scans only measure active brown adipose tissue.4
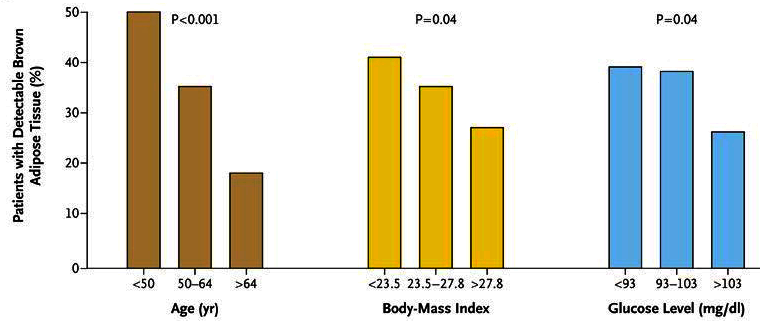
Correlation between the Prevalence of Maximal Activity of Brown Adipose Tissue and Age, Body-Mass Index, and Glucose Level. 5
The exact cause of declining brown fat with age is still a mystery, but is probably connected to the sympathetic nervous system and the thyroid axis. Research data suggests that it may be possible to recruit brown fat throughout a lifetime.
The incidence of obesity, a serious pathology consisting of an imbalance between energy consumption and energy expenditure, increases with age. Brown adipocytes and thermogenesis increase energy expenditure, regulate body weight and reduce the incidence of obesity.6 Obesity and other age related metabolic conditions have been linked to the impairment of brown adipogenic stem cells and cell regeneration.
The loss of brown adipose tissue and metabolic function, including the impairment of mitochondrial biogenesis, has inspired researchers to develop therapies that will increase the generation of brown adipocytes and thermogenic activity. Impaired thermogenesis and regeneration in brown adipocytes may be triggered by neuronal control dysfunction related to the sympathetic nervous system. The loss of active brown adipose tissue may also be exacerbated by alterations in endocrine system signaling.
Targeting Brown Fat Cells using beta-lapachone
Recent research indicates that beta-lapachone is able to convert white adipose tissue into brown fat in mice. The fat browning effect may deliver therapeutic benefits in the treatment of diet induced obesity and age related disease. Beta-lapachone treatment in combination with a high fat diet stimulated the expression of brown adipocyte-specific genes, decreased body weight gain, and mediated higher energy expenditure when compared to a control group.2
Brown fat research has confirmed that an increase in brown adipose tissue levels can reduce the onset of diet induced obesity and other age related diseases. Brown fat generates heat to regulate body temperature when the external environment is cold. Brown fat cells use mitochondria to uncouple oxidative phosphorylation from ATP production, allowing them to generate heat by burning fatty acid energy substrates. It seems clear that individuals with low brown adipose tissue levels are at a higher risk for developing insulin resistance, obesity and cardiovascular disease.7 Brown fat is known to regulate body weight and protect vital organ systems. Among the ways that brown fat regeneration and mitochondrial activity can be targeted are intermittent stress regimens, diet and lifestyle adaptations, exercise programs, drugs, dietary supplements and brown adipose cell transplants.
References:
1: Improving hair growth with Skin remodeling Copper peptides. L Pickart Cosmetics & Medicine (Russia) 2004 July.
2: Beta-lapachone prevents diet-induced obesity by increasing energy expenditure and stimulating the browning of white adipose tissue via down-regulation of miR-382 expression.
WH Choi PMID 2016 May 31;pii:db151423.3: Brown-fat paucity due to impaired BMP signaling induces compensatory browning of white fat.
TJ Schulz et al PMID 2013 Mar 21;495(7441):379-83.doi: 10.1038/nature11943.4: Brown adipose tissue and aging.
V Lecoultre PMID 2011 Jan;14(1):1-6.doi: 10.1097/MCO.0b013e328341221e.5: Identification and Importance of Brown Adipose Tissue in Adult Humans.
N Engl J Med 2009; 360:1509-1517April 9, 2009DOI: 10.1056/NEJMoa08107806: Mechanisms of aging-related impairment of brown adipocyte development and function.
A Graja TJ Schulz Gerontology 2015;61:211-217.DOI:10.1159/000366557).7: Does brown fat protect against diseases of aging?
M Mattson Ageing Res Rev. 2010 Jan;9(1): 69.doi: 10.1016/j.arr.2009.11.004.

Leave a comment